World Congress on Advanced Biomaterials and Tissue Engineering
Theme: Innovations in Biomaterials and Emerging Technologies in Tissue Engineering
Date: October 17-18, 2018
Conference Venue: Rome, Italy
Monday, 15 October 2018
Wednesday, 19 September 2018
Hepatic Tissue Engineering :
The liver has over 500 functions, including protein, carbohydrate, and lipid metabolism; detoxification of endogenous and exogenous compounds; production of bile for digestion; and secretion of many serum proteins (i.e. albumin, coagulation factors). Each year, over 40,000 people die due to liver failure in the US alone, with over 2 million deaths estimated worldwide. Orthotopic liver transplantation is the only proven therapy for liver failure; however, there is a severe shortage of donor organs. Cell-based therapies have been proposed as an alternative to whole organ transplantation, as a temporary bridge to transplantation, and/or an adjunct to traditional therapies during liver regeneration. The three main approaches that have been proposed are: transplantation of isolated hepatocytes, implantable tissue-engineered constructs, and perfusion of blood through an extracorporeal bioartifical liver device containing parenchymal liver cells called hepatocytes. Despite significant investigations into each of these areas, progress has been stymied due to the propensity for isolated hepatocytes to rapidly lose viability and key liver-specific functions upon isolation from the native microenvironment of the liver.

Researchers have used microtechnology tools and biomaterials to synthesize 2D and 3D hepatic microenvironments to study determinants of cell fate and function and then perturb and interrogate these to model human disease. Researchers focused on (i) synthesizing human liver microenvironments for in vitro and in vivo interrogation and (ii) perturbing liver microenvironments with pathogens to model human disease. Some of the notable contributions include the discovery of small molecules that drive proliferation of adult hepatocytes and maturation of stem-cell derived progeny to enable sourcing of human hepatocytes, and the development of the first high-throughput model systems to study hepatotropic pathogens.
Saturday, 15 September 2018
Understanding interactions between Biomaterials and Biological systems using proteomics :

The role that biomaterials play in the clinical treatment of damaged organs and tissues is changing. While biomaterials used in permanent medical devices were required to passively take over the function of a damaged tissue in the long term, current biomaterials are expected to trigger and harness the self-regenerative potential of the body in situ and then to degrade, the foundation of regenerative medicine. To meet these different requirements, it is imperative to fully understand the interactions biomaterials have with biological systems, in space and in time. This knowledge will lead to a better understanding of the regenerative capabilities of biomaterials aiding their design with improved functionalities (e.g. biocompatibility, bioactivity). Proteins play a pivotal role in the interaction between biomaterials and cells or tissues. Protein adsorption on the material surface is the very first event of this interaction, which is determinant for the subsequent processes of cell growth, differentiation, and extracellular matrix formation. Against this background, the aim of the current review is to provide insight in the current knowledge of the role of proteins in cell–biomaterial and tissue–biomaterial interactions. In particular, the focus is on proteomics studies, mainly using mass spectrometry, and the knowledge they have generated on protein adsorption of biomaterials, protein production by cells cultured on materials, safety and efficacy of new materials based on nanoparticles and the analysis of extracellular matrices and extracellular matrix–derived products. In the outlook, the potential and limitations of this approach are discussed and mass spectrometry imaging is presented as a powerful technique that complements existing mass spectrometry techniques by providing spatial molecular information about the material-biological system interactions.

Thursday, 6 September 2018
Magnesium and its alloys as Orthopedic Biomaterials :
As a lightweight metal with mechanical properties similar to natural bone, a natural ionic presence with significant functional roles in biological systems, and in vivo degradation via corrosion in the electrolytic environment of the body, magnesium-based implants have the potential to serve as biocompatible, osteoconductive, degradable implants for load-bearing applications.
Current metallic biomaterials are essentially neutral in vivo, remaining as permanent fixtures, which in the case of plates, screws and pins used to secure serious fractures, must be removed by a second surgical procedure after the tissue has healed sufficiently. Repeat surgery increases costs to the health care system and further morbidity to the patient. Magnesium is an exceptionally lightweight metal. With a density of 1.74 g/cm3, magnesium is 1.6 and 4.5 times less dense than aluminium and steel, respectively. The fracture toughness of magnesium is greater than ceramic biomaterials such as hydroxyapatite, while the elastic modulus and compressive yield strength of magnesium are closer to those of natural bone than is the case for other commonly used metallic implants. Moreover, magnesium is essential to human metabolism and is naturally found in bone tissue.
Wednesday, 5 September 2018
Synthesis, properties, and biomedical applications of gelatin methacryloyl (GelMA) hydrogels :


Gelatin methacryloyl (GelMA) hydrogels have been widely used for various biomedical applications due to their suitable biological properties and tunable physical characteristics. GelMA hydrogels closely resemble some essential properties of native extracellular matrix (ECM) due to the presence of cell-attaching and matrix metalloproteinase responsive peptide motifs, which allow cells to proliferate and spread in GelMA-based scaffolds. GelMA is also versatile from a processing perspective. It crosslinks when exposed to light irradiation to form hydrogels with tunable mechanical properties. It can also be microfabricated using different methodologies including micromolding, photomasking, bioprinting, self-assembly, and microfluidic techniques to generate constructs with controlled architectures. Hybrid hydrogel systems can also be formed by mixing GelMA with nanoparticles such as carbon nanotubes and graphene oxide, and other polymers to form networks with desired combined properties and characteristics for specific biological applications. Recent research has demonstrated the proficiency of GelMA-based hydrogels in a wide range of tissue engineering applications including engineering of bone, cartilage, cardiac, and vascular tissues, among others. Other applications of GelMA hydrogels, besides tissue engineering, include fundamental cell research, cell signaling, drug and gene delivery, and bio-sensing.


Monday, 3 September 2018
Biomaterials and Mesenchymal Stem Cells for Regenerative Medicine :
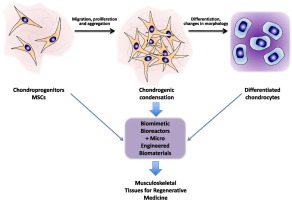
The reconstruction of hard and soft tissues is a major challenge in regenerative medicine, since diseases or traumas are causing increasing numbers of tissue defects due to the aging of the population. Modern tissue engineering is increasingly using three-dimensional structured biomaterials in combination with stem cells as cell source, since mature cells are often not available in sufficient amounts or quality. Biomaterial scaffolds are developed that not only serve as cell carriers providing mechanical support, but actively influence cellular responses including cell attachment and proliferation. Chemical modifications such as the incorporation of chemotactic factors or cell adhesion molecules are examined for their ability to enhance tissue development successfully. E.g. growth factors have been investigated extensively as substances able to support cell growth, differentiation and angiogenesis. Thus, continuously new patents and studies are published, which are investigating the advantages and disadvantages of different biomaterials or cell types for the regeneration of specific tissues. The main focus on biomaterials, including natural and synthetic polymers, ceramics and corresponding composites used as scaffold materials to support cell proliferation and differentiation for hard and soft tissues regeneration. In addition, the local delivery of drugs by scaffold biomaterials.

Thursday, 30 August 2018
Restore, Repair and Regenerate - Aspects and Prospects of Regenerative Medicine and Tissue Engineering :
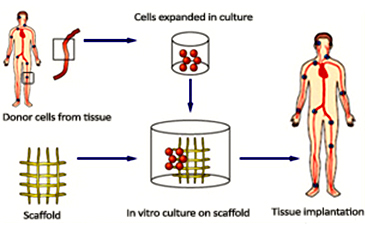
Regenerative medicine and tissue engineering are rapidly growing fields of study that involves in repairing, restoring and regenerating the damaged or lost tissue. They are interdisciplinary fields of science that involves material science, cell biology, biochemistry and general engineering and medicine principles. According to National Institute of Health (NIH), Regenerative Medicine or Tissue Engineering is defined as "rapidly growing interdisciplinary area which involves physical and engineering sciences to develop functional cells, tissues and organs in order to repair, restore or regenerate or to enhance the lost biological function due to injury, abnormalities or ageing".
Components of Tissue Engineering
A general tissue Engineering product contains the following components: Cells, scaffold/biomaterial and biomolecules or signaling molecules. The procedure involves in seeding the suitable cell type on a biocompatible biomaterial often referred as scaffold together with the signaling molecules in order to grow the desired tissue.

Biocompatible materials - The choice of biomaterial used influence the growth, cell differentiation and proliferation in the process of tissue engineering.
Cells - The cells used in the tissue engineering process can be taken from the patient or from the same person to whom the regenerative medicine is applicable (autologous), from another person or donor (allogenic) or the animal cells (xenogenic) and the stem cells.
Wednesday, 29 August 2018
Cell-Biomaterial interaction for construction of synthetic Tissue Microenvironment :

Specific interactions between cell and biomaterials are required to control the cellular functions and for development of a cell (or stem cell) niche. These interactions provide rational designs for construction of specific tissue microenvironment for physiological and pathological conditions.
The design of biomaterials and the sourcing for appropriate cells are two integrated aspects of tissue engineering to construct a tissue implant for clinical applications. During the past decades, many innovative biomaterials with desirable biological and mechanical properties have emerged, while stem cells have been shown to be a promising cell source to differentiate into many cell types. However, the testing of these bioartificial tissue constructs in the clinical trials is far from satisfactory. How microenvironments in the biomaterials regulate cellular signaling pathways and functions, how stem cell-derived target cells respond to extracellular cues presented by the biomaterials, and how implanted tissue constructs interact with host tissues remain to be investigated.
The fundamental cross-talk between a cell and material to provide microenvironmental cues and in understanding the role (and interplay) of the cues in controlling cellular functions including stem cell for cell adhesion, proliferation, migration and differentiation. The information of the interplay between cell and biomaterials would be helpful to guide us in improving our current strategy to refine the tissue constructs for effective tissue repair in regenerative medicine.
Friday, 24 August 2018
Recent Advances in Tissue Engineering Strategies for the Treatment of Joint Damage :

While the clinical potential of tissue engineering for treating joint damage has yet to be realized, research and commercialization efforts in the field are geared towards overcoming major obstacles to clinical translation, as well as towards achieving engineered grafts that recapitulate the unique structures, function, and physiology of the joint. Recent advances in technologies aimed at obtaining biomaterials, stem cells, and bioreactors that will enable the development of effective tissue-engineered treatments for repairing joint damage.
3D printing of scaffolds is aimed at improving the mechanical structure and microenvironment necessary for bone regeneration within a damaged joint. Advances in our understanding of stem cell biology and cell manufacturing processes are informing translational strategies for the therapeutic use of allogeneic and autologous cells. Finally, bioreactors used in combination with cells and biomaterials are promising strategies for generating large tissue grafts for repairing damaged tissues in pre-clinical models. Together, these advances along with ongoing research directions are making tissue engineering increasingly viable for the treatment of joint damage.

Wednesday, 22 August 2018
Nanofiber Scaffold for Utility in Bone Tissue Regeneration :
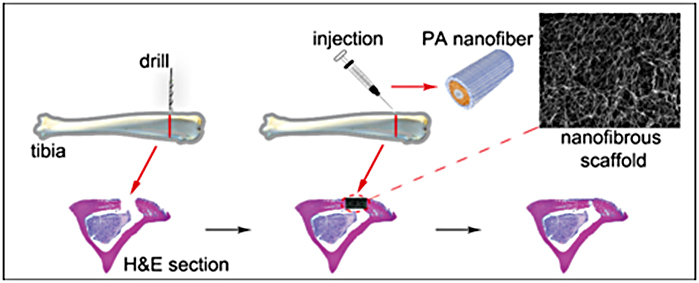
Many variables serve to alter the process of bone remodeling and diminish regeneration including the size and nature of the wound bed and health status of the individual. To overcome these inhibitory factors, tissue engineered osteoconductive scaffolds paired with various growth factors have been utilized clinically. Bone is a highly vascularized and dynamic tissue that has an innate capacity for healing after injury. However, there are many variables that serve to alter the process of bone remodeling that diminish regeneration including the size and nature of the wound bed and chronic medical conditions. To overcome these inhibitory factors, tissue engineered, osteoconductive scaffolds paired with various growth factors have been utilized clinically in orthopedics and craniofacial surgery. However, many limitations still remain with commercially available products (e.g. rhBMP2) which can lead to rampant inflammation associated with injury or clinical intervention, ectopic bone formation, and ultimately graft failure. The ability for a nanofiber scaffold (Talymed), currently approved to augment cutaneous wound healing, to accelerate growth factor (rhBMP2) generated bone healing compared to the traditional absorbable collagen sponge (ACS) delivery system. To assess this healing after craniofacial fracture, 155 adult wild-type mice were randomly arranged in 16 groups by time, 4 and 8 week, and treatment, ACS or Talymed, loaded with control, low, medium or high dosages of rhBMP2. At experimental end points, skulls were subjected to microCT, biomechanical, and histological analysis to assess bone regeneration.
The use of Talymed within the defect site was found to decrease the bone volume, bone formation rate, and alkaline phosphatase positivity compared to ACS/rhBMP2 combinations. Interestingly, the Talymed regenerated bone, although less, was found to have a greater hardness value than that of bone within the ACS groups. However, the difference in bone hardness between scaffolds was not detectable by 8 weeks.

Thursday, 16 August 2018
Advances in Skin Regeneration Using Tissue Engineering
Advances in Skin Regeneration Using Tissue Engineering :
Tissue engineered skin substitutes for wound healing have evolved tremendously over the last couple of years. New advances have been made toward developing skin substitutes made up of artificial and natural materials. Engineered skin substitutes are developed from acellular materials or can be synthesized from autologous, allograft, xenogenic, or synthetic sources. Each of these engineered skin substitutes has their advantages and disadvantages. However, to this date, a complete functional skin substitute is not available, and research is continuing to develop a competent full thickness skin substitute product that can vascularize rapidly. There is also a need to redesign the currently available substitutes to make them user friendly, commercially affordable, and viable with longer shelf life.
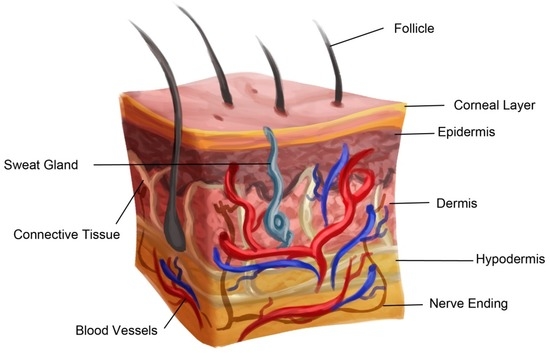
Approaches for Tissue Engineering:
Different strategies, such as injecting growth factors and extracellular matrix, are being adopted towards tissue re-growth and wound healing. Some of the recent strategies are listed below.- Cell Cocultures
- Cultured Epithelial Autografts
- Tissue Engineered Skin Substitutes
Tissue engineered skin substitute preparation. Bold lines indicate cell type for tissue engineered substitute and dotted lines indicate cell source
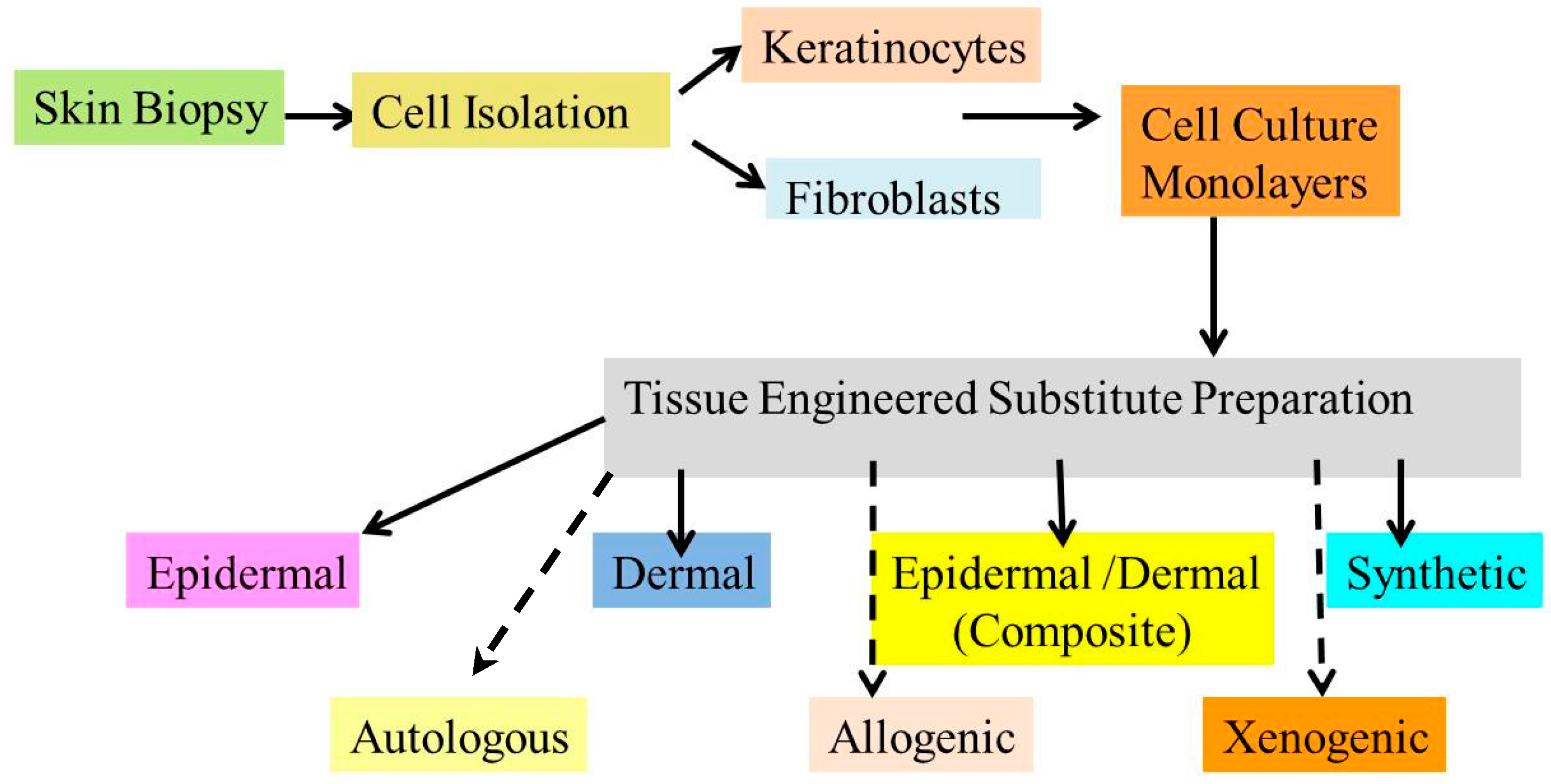
Types of Skin Substitutes:
- Acellular Skin Substitutes
- Cellular Allogenic Skin Substitutes
- Cellular Autologous Skin Substitutes
- Commercially Available Skin Substitutes
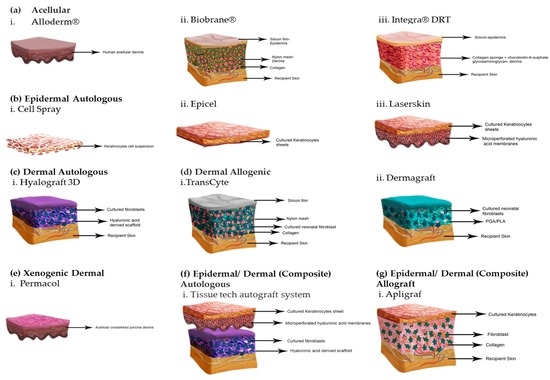
Tissue engineered skin substitutes
Tuesday, 14 August 2018
Biomaterials and Therapeutic Applications :
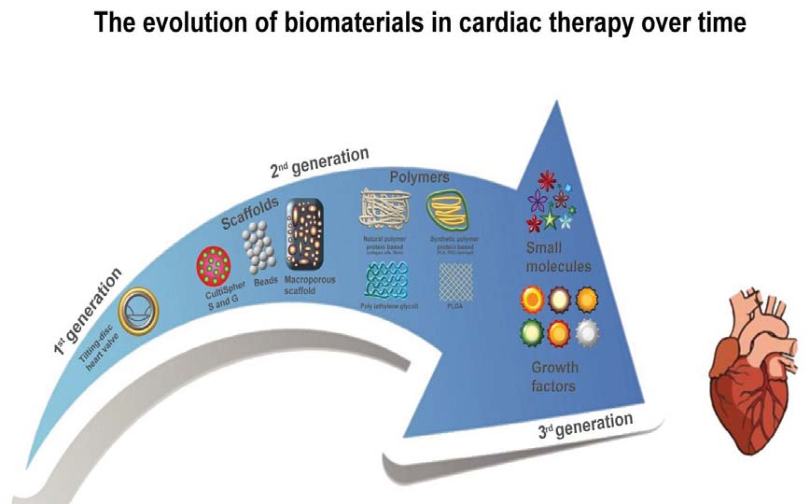
Millions of patients worldwide have benefited from technological innovation from biomaterials. Yet, while life expectancy continues to increase, organ failure and traumatic injury continue to fill hospitals and diminish the quality of life. A number of organic and inorganic, synthetic or natural derived materials have been classified as not harmful for the human body and are appropriate for medical applications. These materials are usually named biomaterials since they are suitable for introduction into living human tissues of prosthesis, as well as for drug delivery, diagnosis, therapies, tissue regeneration and many other clinical applications. Advances in understanding disease and tissue regeneration combined with increased accessibility of modern technology have created new opportunities for the use of biomaterials in unprecedented ways. Materials can now be rapidly created and selected to target specific cells, change shape in response to external stimulus, and instruct tissue regeneration. Recently, nanomaterials and bioabsorbable polymers have greatly enlarged the fields of application of biomaterials attracting much more the attention of the biomedical community.
One such example is use of Biomaterials in Cardiac Therapy is mentioned below.
Monday, 13 August 2018
Synthesis and surface engineering of iron oxide Nanoparticles for Biomedical applications :
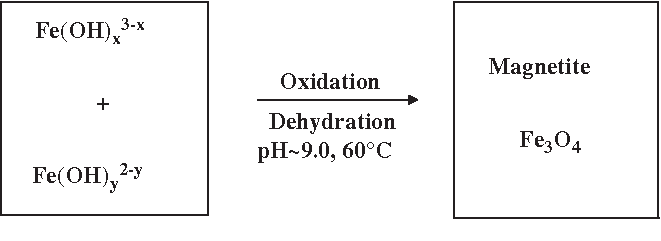
Superparamagnetic iron oxide nanoparticles (SPION) with appropriate surface chemistry have been widely used experimentally for numerous in vivo applications such as magnetic resonance imaging contrast enhancement, tissue repair, immunoassay, detoxification of biological fluids, hyperthermia, drug delivery and in cell separation, etc. All these biomedical and bioengineering applications require that these nanoparticles have high magnetization values and size smaller than 100 nm with overall narrow particle size distribution, so that the particles have uniform physical and chemical properties. In addition, these applications need special surface coating of the magnetic particles, which has to be not only non-toxic and biocompatible but also allow a targetable delivery with particle localization in a specific area. To this end, most work in this field has been done in improving the biocompatibility of the materials, but only a few scientific investigations and developments have been carried out in improving the quality of magnetic particles, their size distribution, their shape and surface in addition to characterizing them to get a protocol for the quality control of these particles. Nature of surface coatings and their subsequent geometric arrangement on the nanoparticles determine not only the overall size of the colloid but also play a significant role in biokinetics and biodistribution of nanoparticles in the body. The types of specific coating, or derivatization, for these nanoparticles depend on the end application and should be chosen by keeping a particular application in mind, whether it be aimed at inflammation response or anti-cancer agents. Magnetic nanoparticles can bind to drugs, proteins, enzymes, antibodies, or nucleotides and can be directed to an organ, tissue, or tumour using an external magnetic field or can be heated in alternating magnetic fields for use in hyperthermia.

Subscribe to:
Posts (Atom)
