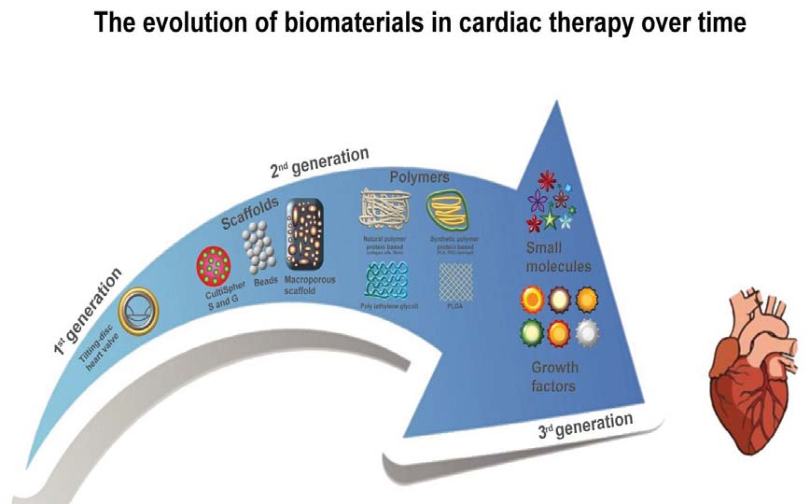
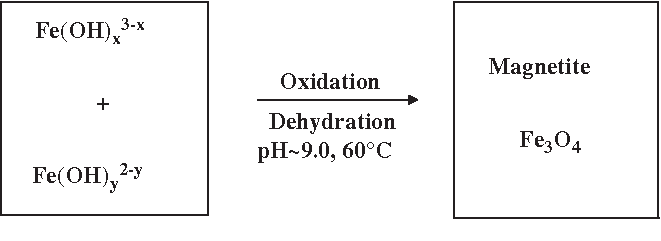
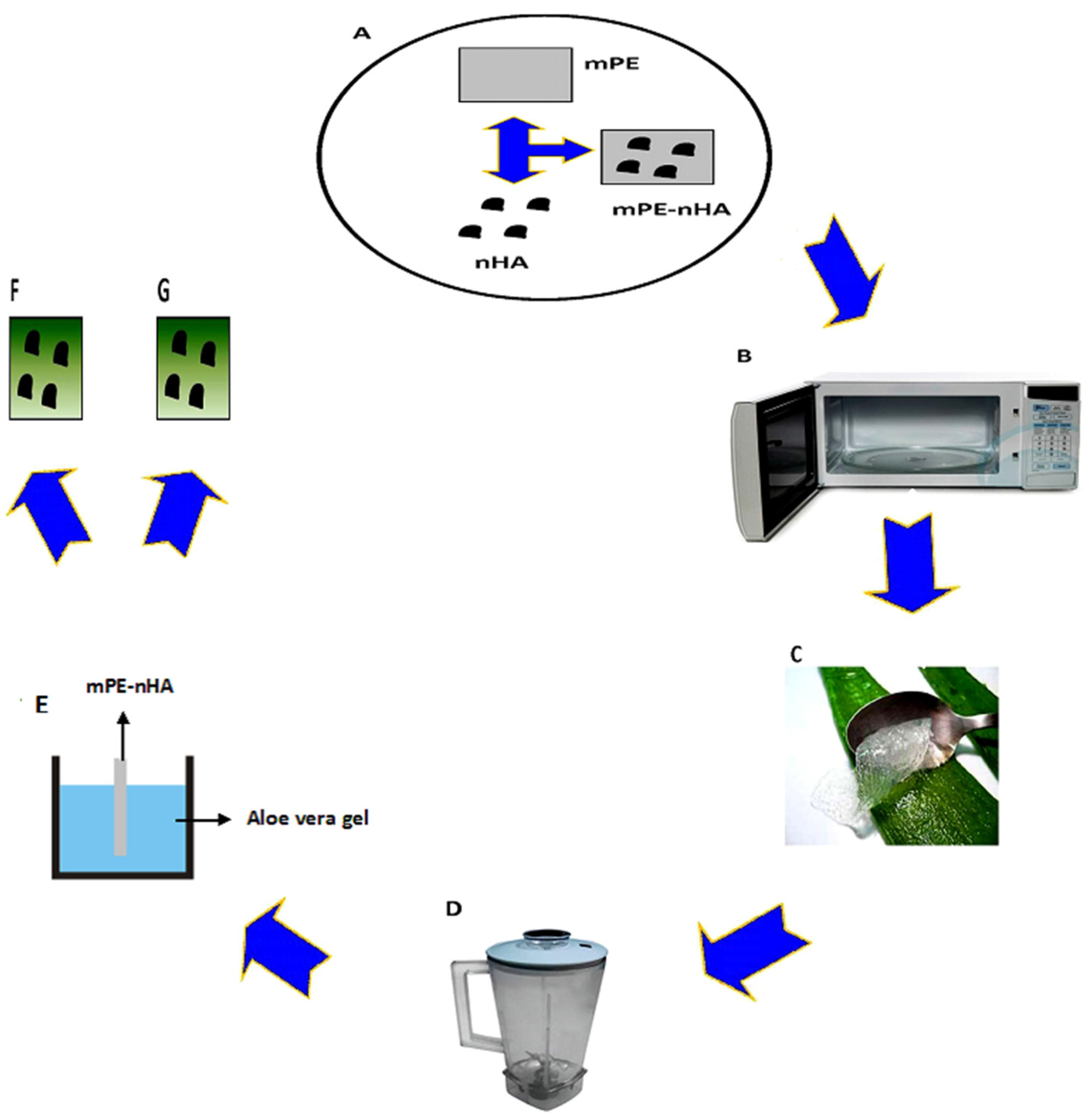
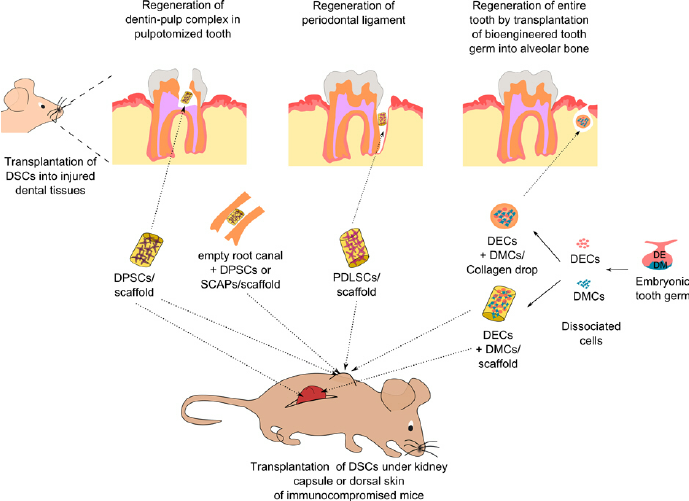
Restore, Repair and Regenerate - Aspects and Prospects of Regenerative Medicine and Tissue Engineering :
Regenerative medicine and tissue engineering are rapidly growing fields of study that involves in repairing, restoring and regenerating the damaged or lost tissue. They are interdisciplinary fields of science that involves material science, cell biology, biochemistry and general engineering and medicine principles. According to National Institute of Health (NIH), Regenerative Medicine or Tissue Engineering is defined as "rapidly growing interdisciplinary area which involves physical and engineering sciences to develop functional cells, tissues and organs in order to repair, restore or regenerate or to enhance the lost biological function due to injury, abnormalities or ageing".
Components of Tissue Engineering
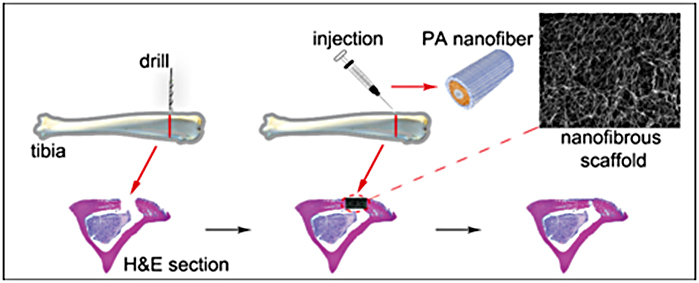
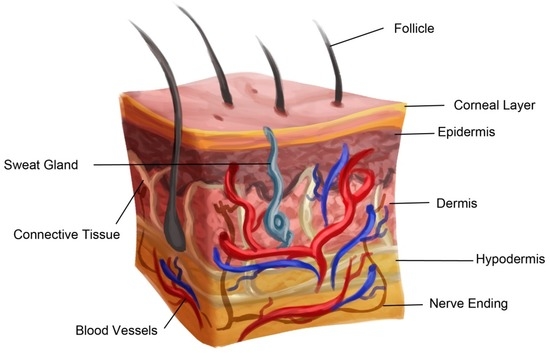
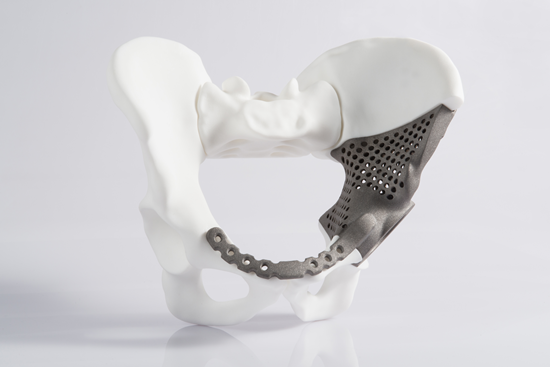
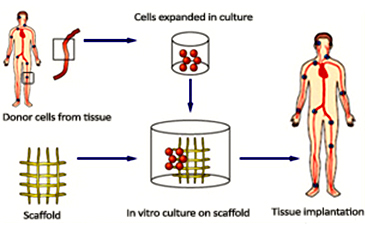
A general tissue Engineering product contains the following components: Cells, scaffold/biomaterial and biomolecules or signaling molecules. The procedure involves in seeding the suitable cell type on a biocompatible biomaterial often referred as scaffold together with the signaling molecules in order to grow the desired tissue.

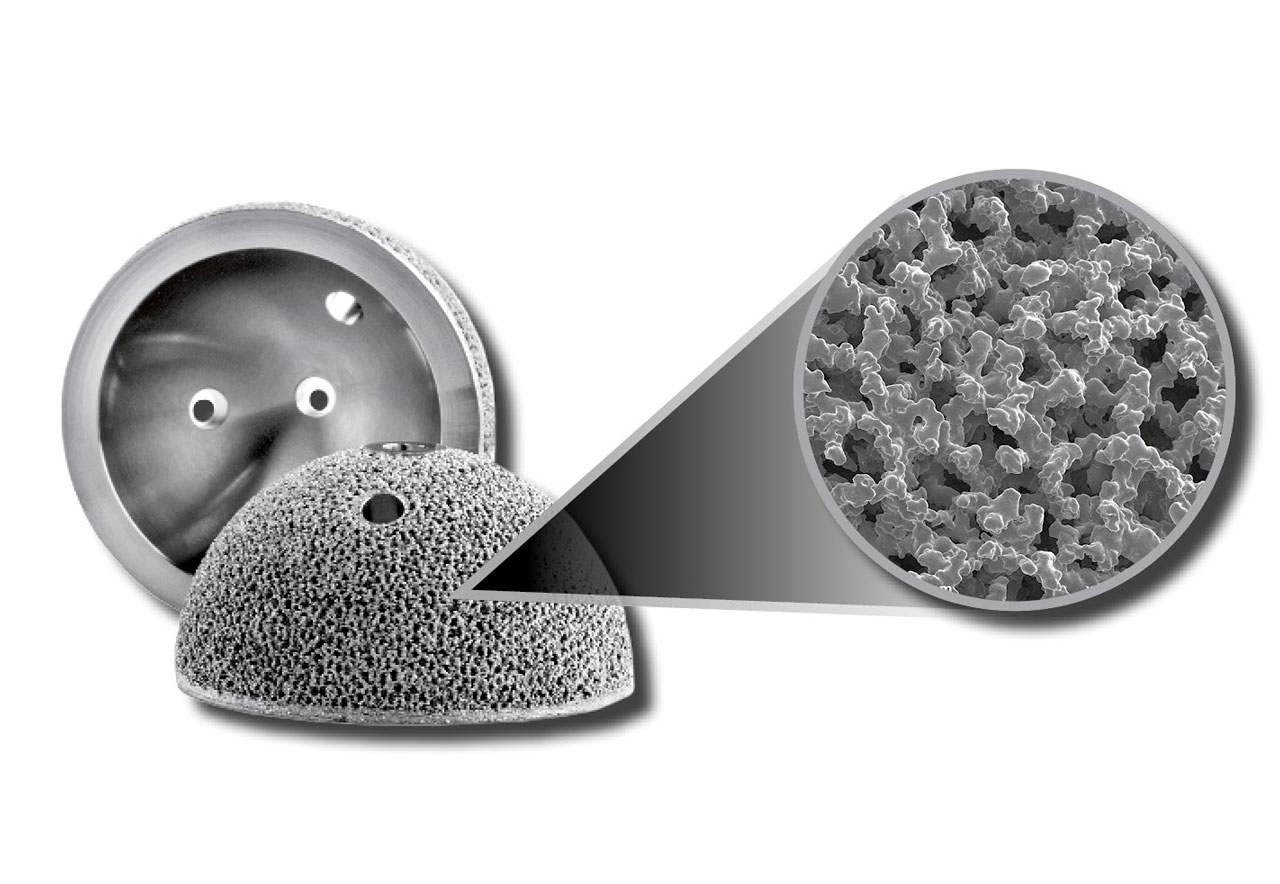
Biocompatible materials - The choice of biomaterial used influence the growth, cell differentiation and proliferation in the process of tissue engineering.
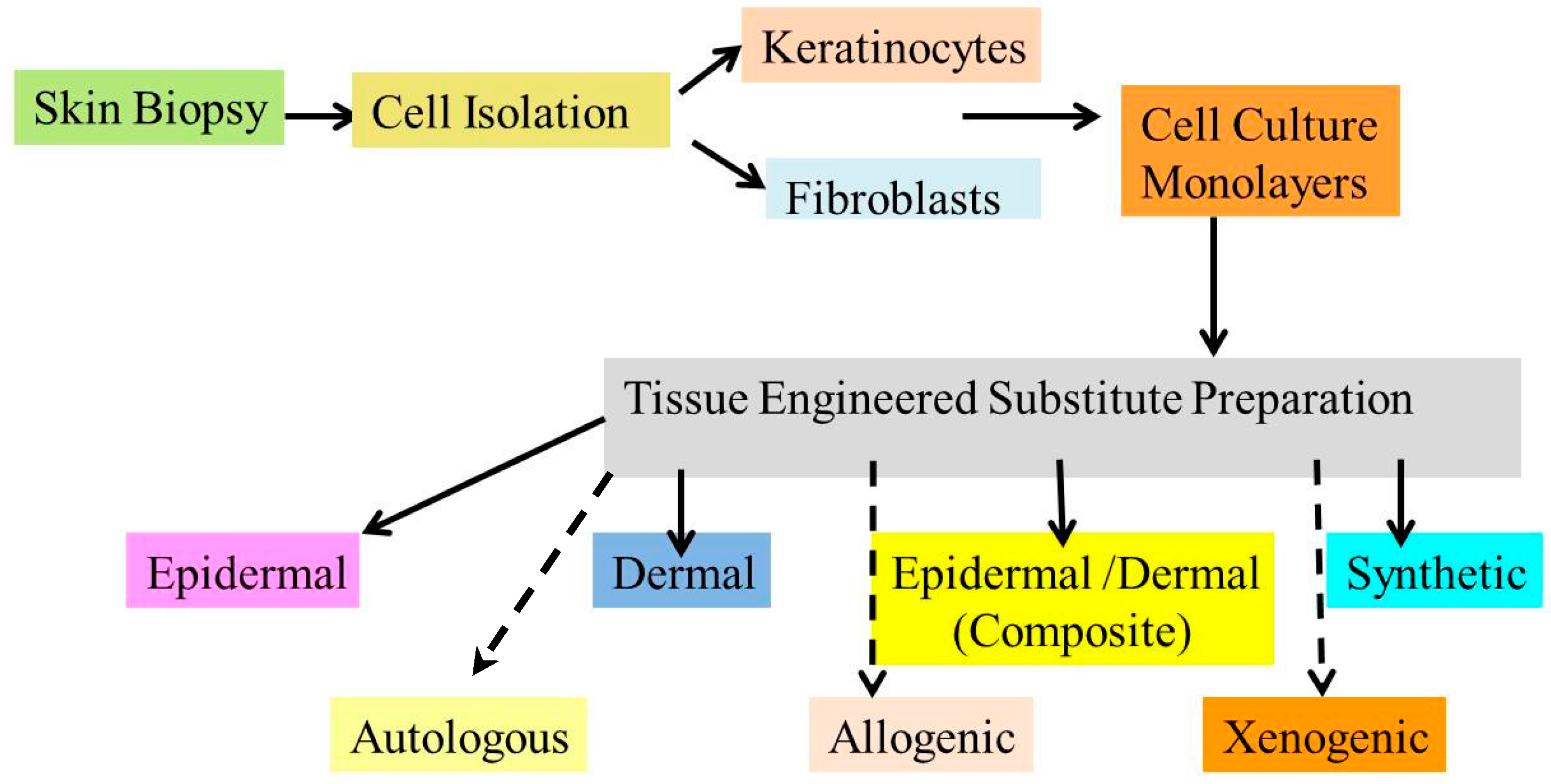
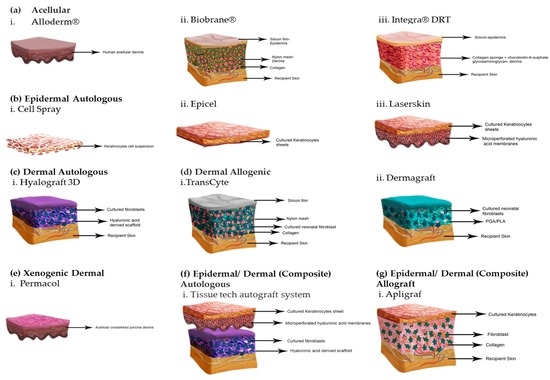
Cells - The cells used in the tissue engineering process can be taken from the patient or from the same person to whom the regenerative medicine is applicable (autologous), from another person or donor (allogenic) or the animal cells (xenogenic) and the stem cells.